There are no projects in the garbage can.
Our main objective is to dissect the molecular pathways through which diet modulates biological processes involved in health and disease. Diet is the primary source of nutrients, and there is strong evidence that both the type and amount of food we consume have a major impact on disease risk and progression. However, the molecular mechanisms underlying these effects remain incompletely understood. Addressing this question is essential to determine how specific dietary factors modulate distinct biological signaling pathways that impact health and disease.
A central focus of our work is the study of ketosis and its impact on human physiology. Ketosis is a conserved metabolic state triggered by carbohydrate restriction and characterized by increased levels of circulating ketone bodies, particularly β-hydroxybutyrate (BHB). While traditionally viewed as an alternative energy source during glucose scarcity, BHB is now recognized as a signaling metabolite with broad biological effects. Despite growing interest in the use of ketogenic interventions in conditions such as obesity, diabetes, cancer, inflammation, and aging, the mechanisms through which BHB and ketosis influence whole-body physiology remain poorly defined. This lack of mechanistic understanding, together with variable and conflicting clinical outcomes, limits their safe and effective therapeutic application.
Our recent work has uncovered a previously unrecognized metabolic pathway in which the cytosolic enzyme CNDP2 conjugates BHB with amino acids to form a new class of metabolites, the BHB-amino acids. These endogenous metabolites are present in both mice and humans, and their circulating levels are dynamically regulated by ketosis. We have shown that BHB-amino acids are bioactive metabolites that suppress appetite in mice, therefore linking ketosis to weight control in obesity.
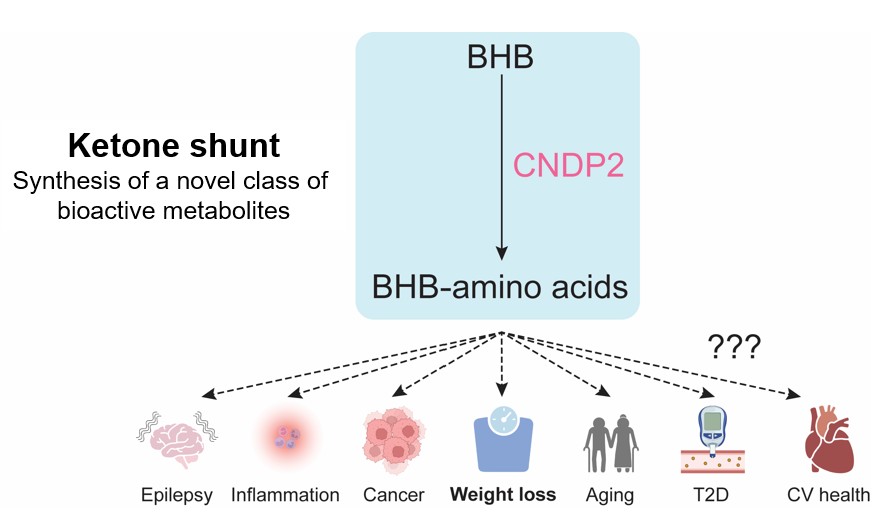
The identification of this ketone shunt pathway opens the possibility to revisit long-standing questions in ketone biology from a new perspective, asking what other mechanisms of ketosis we may have missed by not recognizing these metabolites and their pathway earlier. The next challenge is to define the physiological context/s in which this pathway operates and assess its relevance beyond obesity.
Our current work focuses on understanding the biological functions of these metabolites in physiological and pathological contexts in which ketosis or BHB have been previously implicated, particularly inflammation and cancer. More broadly, our goal is to investigate and de-orphanize pathways and metabolites that mediate the effects of diet on human health.
Our research is embedded within the Personalised Medicine area and has both therapeutic and biomarker discovery applications. At the same time, it contributes to the Functional Genomics area through the discovery and mechanistic characterisation of novel diet-regulated metabolic pathways involved in inflammation and cancer.
Our research lines:
We integrate biochemistry with mass spectrometry-based metabolomics for discovery. This is complemented by functional validation in cellular and animal models, as well as by translational studies in human samples through clinical collaborations.
Bibliography related to the group research lines
Moya-Garzon, M. D.; Wang, M.; Li, V. L.; Lyu, X.; Wei, W.; Tung, A. S.-H.; Raun, S. H.; Zhao, M.; Coassolo, L.; Islam, H.; Oliveira, B.; Dai, Y.; Spaas, J.; Delgado-Gonzalez, A.; Donoso, K.; Alvarez-Buylla, A.; Franco-Montalban, F.; Letian, A.; Ward, C. P.; Liu, L.; Svensson, K. J.; Goldberg, E. L.; Gardner, C. D.; Little, J. P.; Banik, S. M.; Xu, Y.; Long, J. Z. A β-Hydroxybutyrate Shunt Pathway Generates Anti-Obesity Ketone Metabolites. Cell 2025, 188 (1), 175-186.e20. https://doi.org/10.1016/j.cell.2024.10.032.
File number: RYC2024-048926-I
Principal Investigator: Dr. Maria Dolores Moya Garzon
Funder: Ministerio de Ciencia, Innovación y Universidades (Agencia Estatal de Investigación)
Execution period: 04/2026 – 03/2031










